Significant changes in the Medical Device Regulation and their Implication for Manufacturers
It is important to note that the Regulation has binding legal force throughout the EU and becomes effective simultaneously in all the Member States.
Conformance with the MDR is a significant business challenge that must be overcome in order to enjoy continued access tothe EU market. To meet this challenge manufacturers must be proactive and begin preparing now.
Vigilance and Post Market Surveillance (PMS)
Manufacturers need to review their procedures for PMS and ensure that the responsibility for the provision of this additional data and associated support is clearly established.
Executive support and proactive programme management of MDR implementation is a prerequisite for a manufacturer’s success in this regard.
Scope of «VigilanX»
Brief overview
OVERVIEW
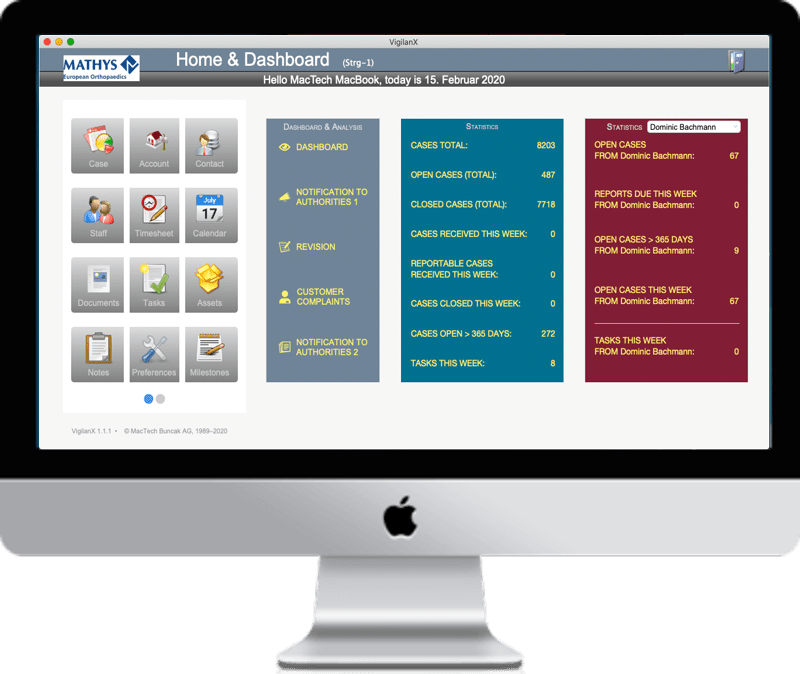
Dashboard
Panel
Panel
View your top risks, management actions and graphs in the snapshot dashboard view
RISKMANAGMENT
Identify
Risks
Risks
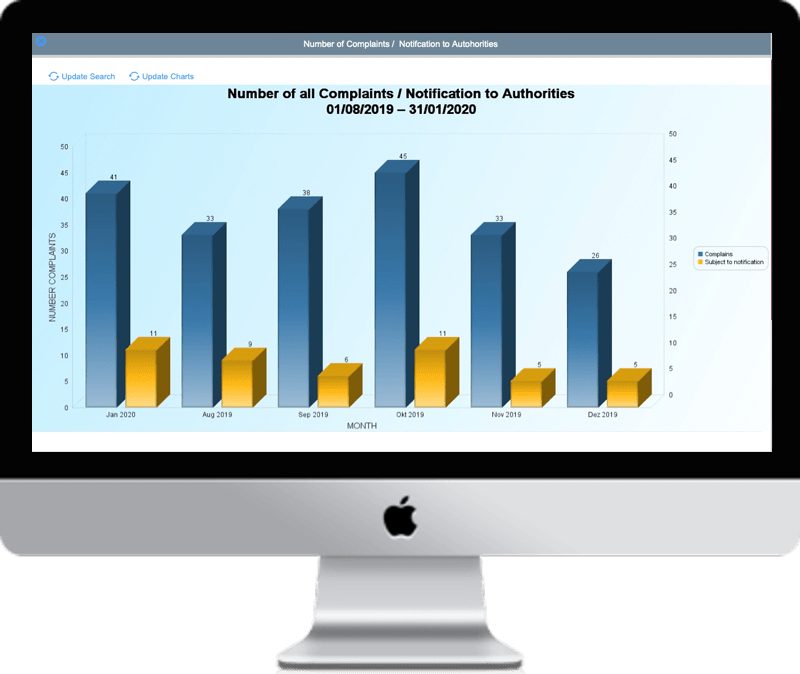
Quickly identify and report on all your data security risks in time

DATA
Data
Assessments
Assessments
Conduct data protection impact assessments easily and consistently
COMPLIANCE
Demonstrate
Compliance
Compliance
Demonstrate your compliance efforts with fast and comprehensive reporting
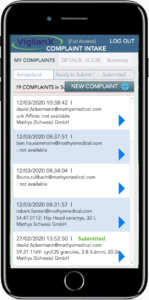
DATA ENTRY
At the customer’s premises
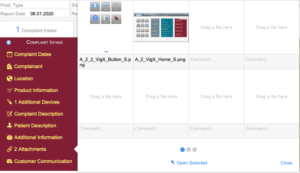
Field staff can record complaints (iPhone Plus or iPad) directly at the customer’s premises, take pictures and log them by using VigilanX-WEB
Meet The Ultimate Team
A Part of our staff is at your disposal
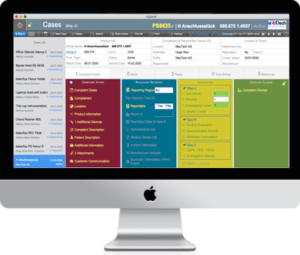
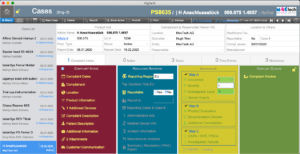
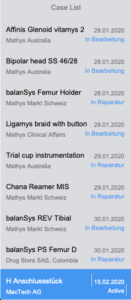
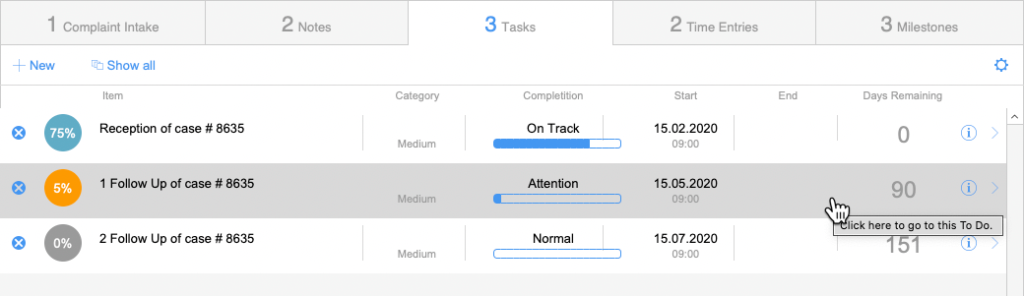
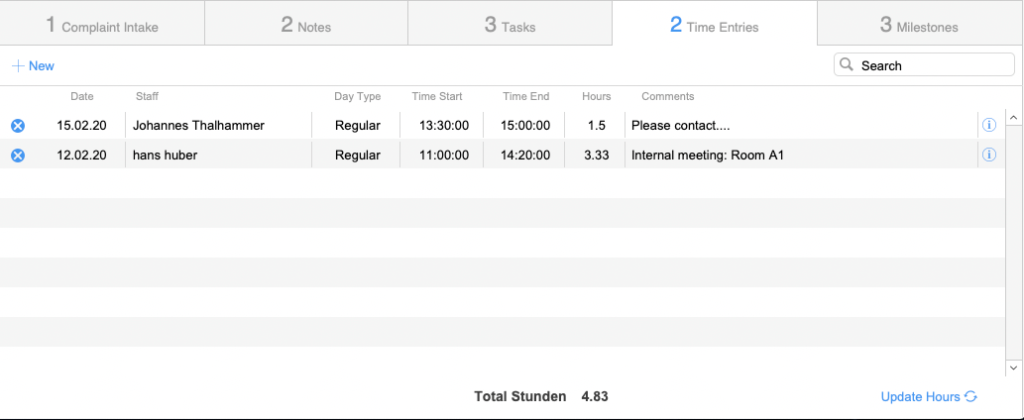
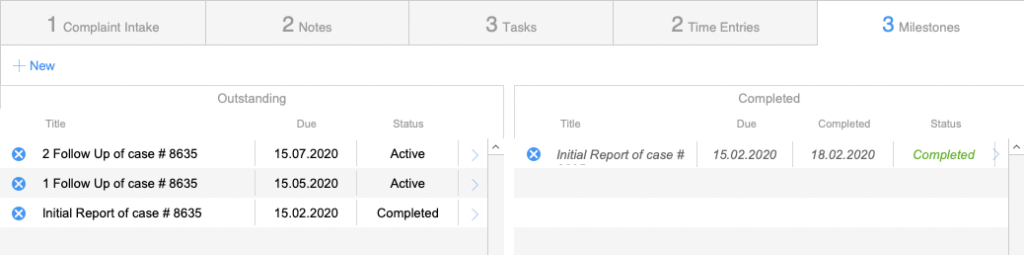
Case Tracking
Overview and functions
Home & Dashboard
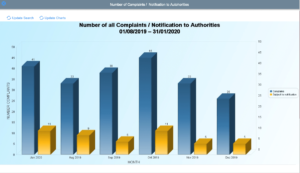
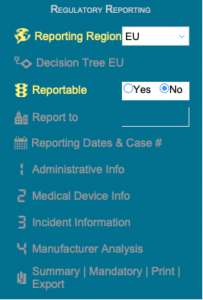
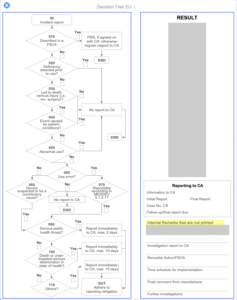
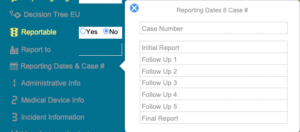
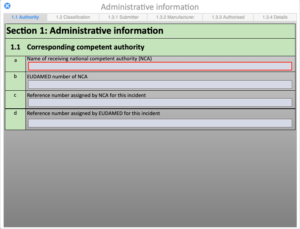
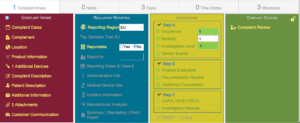
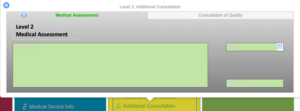
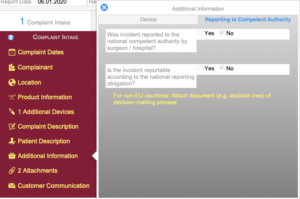
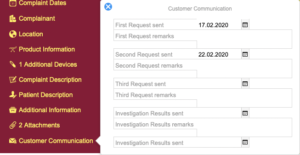
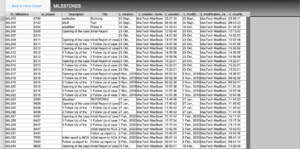
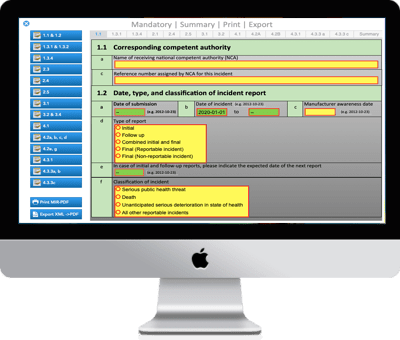
Regulatory Reporting
Case / Overview
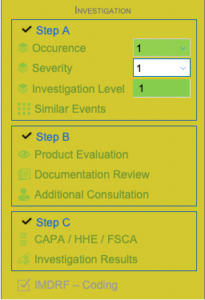
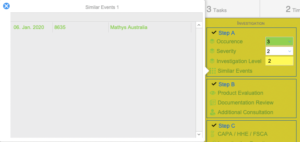
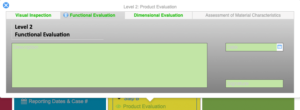
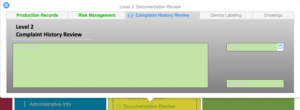
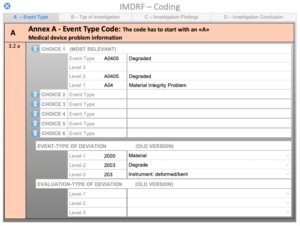
Investigation
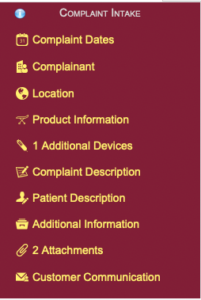
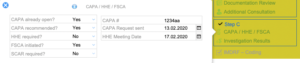
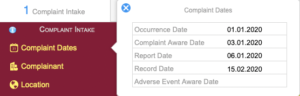
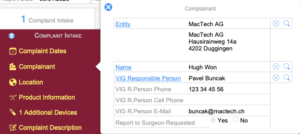
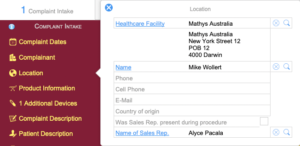
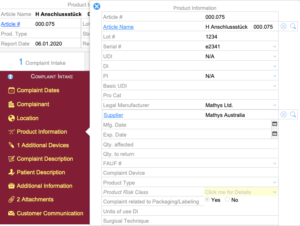
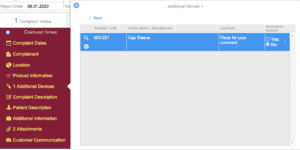
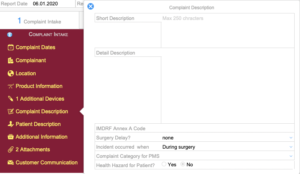
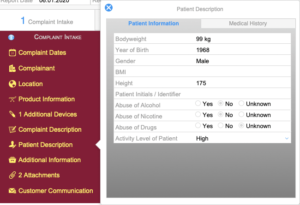
Complaint Intake
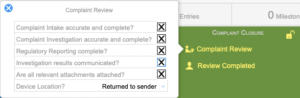
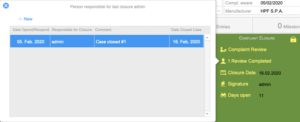
Complaint Closure
Extensions
Add-ons
Case / Overview
Modules
Enhancement of functionality
Home & Modules
Contact
Documents
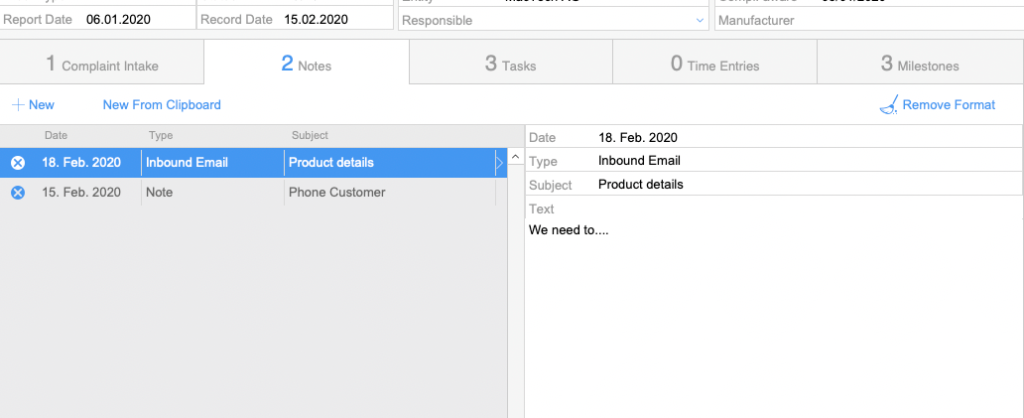
Notes
Case
Staff
Task
Milestones
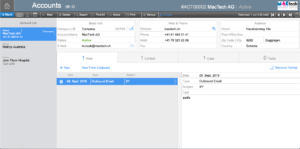
Account
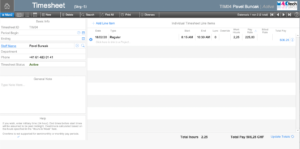
Timesheet
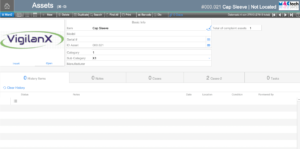
Assets
Preferences
Testimonial
What our customers say
The complexity is extremely well implemented
ky78
Ultimate Customer
The separation between headquarters and branches is optimally solved with the two separate modules
ky78
Ultimate Customer